436
Views & Citations10
Likes & Shares
GENERAL BACKGROUND
Non-aqueous or
organic solvents such as n-butyl chloride (CH3-CH2-CH2-CH2-Cl)
(BuCl), etc., are usually employed in several studies to generate free ions and
radicals of associated solutes. In this the radiolytically generated reactive
species (ions and radicals of organic solvents) involve actively in electron
transfer (ET) processes [1-5]. The transient species produced during radiolysis of neat BuCl are: the parent
radical cations (BuCl•+),
butene radical cations (Bu•+)
and butyl radicals (Bu•)
[2]. The BuCl•+ react through following reactions to produce radical cations and
radicals of solute molecules (SH):
SH + BuCl•+ à SH•+ + BuCl (2)
(SH contains (>N)2C=S moiety)
SH•+ undergoes deprotonation leading
to S•,
SH•+
à S• + H+ (3)
SH + BuCl•+ à S• + BuCl + H+ (4)
Bu•+ has very low gas phase ionization
potential (~1 eV) with respect to BuCl•+ (gas phase IP(BuCl)=10.7 eV), hence not considered [1,2,6] for ET reactions under
the study. Furthermore, the radicals such as Bu• reactions
are well differentiated from radical cations upon O2 addition into
the systems, where the Bu• reacts readily with O2 forming
peroxides [7].
On the other
hand, aqueous solution continues for transient chemistry studies, in which
different methods such as thermal, photolysis, radiolysis and sonolysis are
employed for transients’ generations. The use of radiolysis (with g-ray and e-
beam irradiation) or ultra sound permits solute transient generation indirectly
via the reactions of primary solvent derived redox radicals/ions with solute
molecules. Moreover, in all cases the formation of solute radical cations
undergoes rapid deprotonation limiting other associate chemical investigations.
Hence for such studies either organic solvents (as discussed above) or in
gaseous and frozen matrices are beneficial. Extensive studies on transient
acidity (pKa) and their
chemical reactivity based on kinetic parameters such as redox potentials,
reaction rate constants, etc. are generally unattainable because of the use of
different environment. In this context the use of aqueous acid (HClO4
or H2SO4) has proved to be a useful media for radiation
induced generation and study of protonated transient species conveniently.
[8,9].
In this review
our aim is to bring together the three solvent systems: organic solvent (BuCl),
normal aqueous solutions (pH 0-14) and highly acidic media (pH<0) for
radiation chemical studies on transient species (radical cations and radicals)
wherein thiourea (TU) is used as a model compound, which contains (>N)2C=S
moiety.
DISCUSSION
The pulse
radiolysis experimental set up for radiation chemical study available in the
institute has been reported previously [10] wherein 7 MeV electron beam of
different pulse durations (for example 200 ns; dose rate 40 Gy/pulse,
determined as described elsewhere [11]) was used for sample irradiation. The
dose per pulse in organic medium was calculated considering density of medium
(density of BuCl=0.886 kg L-1) [12] as explained before [13] and
found 35 Gy/pulse in BuCl for 40 Gy/pulse in aqueous systems. Nevertheless, in
aqueous medium, the primary species generated because of the interaction of
ionizing radiation (e--beam) in picosecond time and diffused
homogeneously throughout the medium within 0.1 ms time are: eaq-,
·OH, H·, H2, H2O2, H3O+.
[13] Amongst these, eaq- and H· are reducing and ·OH
is oxidizing species. The molecular species (H2
and H2O2) are
less reactive as
compared to
Furthermore, the generation of SO4•- (an oxidizing radical) during
radiolysis of aqueous H2SO4 is well known [8,9] which
possesses broad absorption band with an absorption maximum (lmax) at
450 nm. The reactions taking place are:
H2O •OH,
H•, H2O2•+/HO2•,
eaq- (6)
•OH + H2SO4/HSO4-
¾® SO4•-
(7)
•OH + SO42- ¾® SO4•-
+ OH- (8)
The H2O2•+/HO2•
(pKa = 1.2) [14] may also
act as oxidant. Under highly acidic conditions H• remains the only
reducing radical produced through eaq- + H3O+
¾® H•. However, in oxygenated aqueous H2SO4
during radiolysis (•H + O2 ¾® HO2•), SO4•- remains
only as reactive oxidizing species to interact with TU, and subsequently
generates TU radical
cations. The concentration of •OH vary with acid strengths and in
acid solvents these radicals (SO4•-
and •OH)
mostly represented as total oxidising radicals [9].
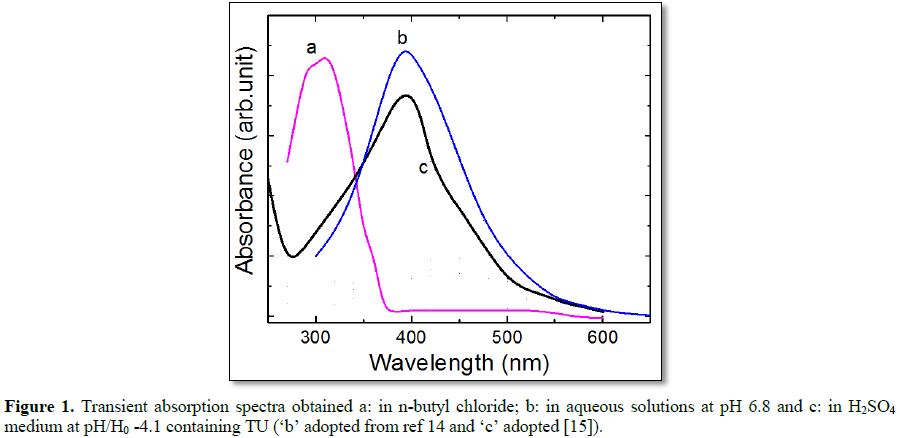
Figure 1 spectrum ‘a’ represents the
transient absorption spectrum obtained in electron pulse irradiated Ar-purged
0.5 mM TU solutions in BuCl, which exhibits a lmax around 300 nm. The absorbance at 300 nm
increases with time leading to the formation of TU radical with slow growth.
This is formed probably due to ET reaction between BuCl•+ and TU via deprotonation (reaction 3), as the growth of intermediate
starts from minimal absorbance value (first component the TU radical cation) immediately after the
electron pulse to
high absorbance (2nd component the TU radical) at 25 ms after the electron
pulse. This is
concluded based on the similar
studies with TU but in presence of ethanol (a
radical cation scavenger) where identical yields of TU radical have been
observed. Under the study because of the limitation of experimental facility the radical cation of solvent was not
observed.
Similarly, the
pulse radiolysis of N2O-saturated aqueous solution of TU at pH 6.8
yielded an intermediate, which exhibits a strong absorption with λmax
at 400 nm (spectrum ‘b’ in Figure 1).
This species as discussed above undergoes deprotonation rapidly resulting TU
radical. Moreover, this radical reacts with parent TU to yield dimer type
species only in basic media (pH 11.9), which exhibits absorption at 510 nm [15].
In the same
way, the transient absorption spectra
reported in electron pulse irradiated aerated 1 mM TU solutions at pH/Ho = -4.1 [16] is compared in Figure
1, in which the spectrum ‘c’ exhibits λmax at 400 nm. This curve is quite similar to
the peak obtained at pH 6.8 (spectrum ‘b’). The rate constant for SO4•- reaction with TU has been reported to be 2.4 ´ 108 dm3 mol-1
s-1 which is rather slow as compared to •OH
radical reactions (1.2 ´ 1010 dm3
mol-1 s-1) [15]. The intermediate has been designated as
TU radical cation, as its decay follows first order kinetics. It is noteworthy
to note that at pH/H0 < -5.0 HSO4, a protonated
form of SO4•-
takes part in oxidation reactions with low reactivity. Hence, >-5.0 Ho
is found to be better for SO4•- reactions study.
It is reported that the yield and the
formation kinetics of the TU transients with respect to absorption at 400 nm depend on pH/H0
of the experimental solutions. The variation of absorbance value with pH/H0
reveals the existence of acid – base conjugate forms of the TU transients and
the pKa values -0.32 and -4.33 has been reported elsewhere [16]. TU possesses better stability
at high acid strength within the present experimental conditions. The
intermediate species responsible for 400 nm absorption decays by following
first order kinetics. The half-life time of TU radical cation increases with acid strength significantly [16]. In high strength acid medium 510 nm light absorbing
intermediate species has not been generated
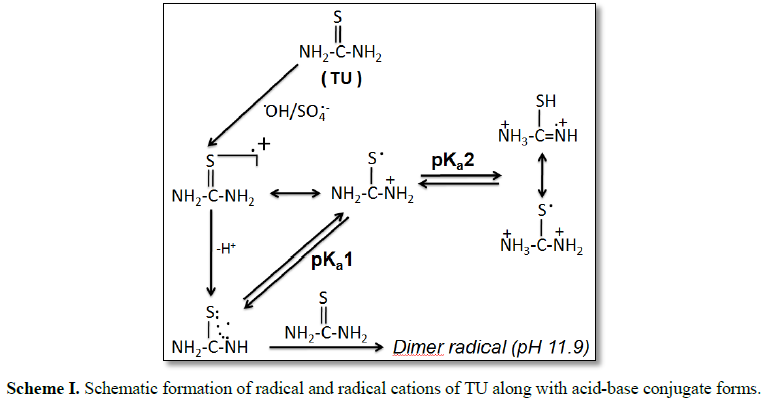
revealing that the reaction for the generation of dimer radical type species are pH selective (Scheme 1).
CONCLUSION
In this
presentation we have demonstrated the effect of experimental media on the
radiolytic generation and stability of radical and radical cations of TU. The
difference in absorption peak/spectra in organic and aqueous media is due to
the difference in polarity of the solvent/solutions suggesting a strong
interaction of the radical cation/radical with the solvent media. Moreover, a
series of TU derivatives have been studied pulse radiolytically in above
mentioned three media [17-21] in which kinetics and stability of their radical
and radical cations differs significantly.
1. Brede
O, Naumov S (2010) Charged particle and photon interactions with matter. Eds.
Hatano Y, Katsumura Y, Mozumdar A. CRC Press: Boca Raton.
2. Mehnert
R, Brede O, Naumann W (1982) Charge transfer from the solvent radical cation to
solutes studied in pulse‐irradiated liquid n‐butyl chloride. Ber Bunsenges Phys
Chem 86: 525-529.
3. Gaebert
C, Mattay J, Toubartz M, Steenken S, Müller B, et al. (2005) Radical cations of
phenyl-substituted aziridines: What are the conditions for ring opening?
Chemistry 11: 1294-304.
4. Parker
VD (1998) Radical and polar reactivity of radical ions in solution. A new look
at an old problem. Acta Chem Scand 52: 145-153.
5. Kim
BD, Shim SC, Ichinose N, Tojo S, Majima T (2001) Formation and properties of
enzyme radical cation. J Photosci 8: 67-69.
6. Lomoth
R, Naumov S, Brede O (1999) Genuine pyrimidine radical cations generated by
radical-induced electron transfer to butyl chloride or acetone parent-ions. J
Phys Chem A 103: 2641-2648.
7. Dey
GR, Hermann R, Naumov S, Brede O (1999) Encounter geometry determines product
characteristics of electron transfer from 4-hydroxythiophenol to n-butyl
chloride radical cations. Chem Phys Lett 310: 137-144.
8. Jiang
PY, Katsumura Y, Nagaishi R, Domae M, lshikawa K, et al. (1992) Pulse
radiolysis study of concentrated sulfuric acid solutions, formation mechanism,
yield and reactivity of sulphate radicals. J Chem Soc Faraday Trans 88:
1653-1658.
9. Das
TN (2008) Radiation induced redox chemistry in aqueous-H2SO4
solvent, in radiation induced free radical chemistry in homo and heterogeneous
media. Dey GR (Eds). Research Signpost: Kerala, India, pp: 39-65.
10. Guha
SN, Moorthy PN, Kishore K, Naik DB, Rao KN (1987) One-electron reduction of
thionine studied by pulse radiolysis. Proc Indian Acad Sci (Chem Sci) 99:
261-271.
11. Buxton
GV, Stuart CR (1995) Re-evaluation of the thiocyanate dosimeter for pulse
radiolysis. J Chem Soc Faraday Trans 91:
279-281.
12. Spinks JWT, Woods RJ (1990) An introduction to radiation chemistry. 3rd
Edn, John Wiley: New York.
13. Sehested
K, Rasmussen OL, Fricke H (1968) Rate constants of OH with HO2, O2-
and H2O2+ from hydrogen peroxide formation in
pulse-irradiated oxygenated water. J Phys Chem 72: 626-631.
14. Wang
WF, Schuchmann MN, Schuchmann HP, Knolle W, Sonntag JV, et al. (1999) Radical
cations in the OH radical induced oxidation of thiourea and tetramethyl
thiourea in aqueous solution. J Am Chem Soc 121: 238-245.
15. Dey
GR (2013) A comparative study of radical cations of thiourea, thiosemicarbazide
and diethylthiourea in aqueous sulphuric acid media using pulse radiolysis
technique. J Phys Org Chem 26: 927-932
16. Dey
GR, Naik DB, Kishore K, Moorthy PN (1994) Kinetics and spectral characteristics
of transients formed in the pulse radiolysis of phenylthiourea in aqueous
solutions. Radiat Phys Chem 43: 365-369.
17. Dey
GR, Naik DB, Kishore K, Moorthy PN (1994) Nature of the transient species
formed in pulse radiolysis of some thiourea derivatives. J Chem Soc Perkin
Trans 2: 1625-1629.
18. Dey
GR, Naik DB, Kishore K, Moorthy PN (1995) Kinetic and spectral properties of
intermediates of 2-mercaptobenzimidazole formed in pulse radiolysis. Res Chem
Intermed 21: 47-58.
19. Dey
GR (2019) Effect of phenyl moiety on the formation of radicals and radical
cations of thioamides in n-butyl chloride: A pulse radiolysis study. J Chem Sci
131: 1-7.
20. Dey
GR (2015) Radicals and radical cations of thioacetamide and thiobenzamide in
aqueous sulphuric acid media. Res Chem Intermed 41: 831-843.
21. Dey
GR (2014) Effect of open and cyclic (>N)2C=S moiety in
phenylthiourea and 2-mercapto-benzimidazole on reactivity of •OH and SO4•-
in aqueous sulphuric acid media. SOP Trans Phys Chem 1: 41-52.